Comparing Lithium Extraction Technologies: IBAT DLE vs Other DLE Methods
From sustainability to cost to recovery rate, IBAT DLE delivers superior results.
In the world of brine recovery, DLE–or Direct Lithium Extraction–offers significant advantages over conventional methods. Brine is where lithium extraction will be focused in the coming years–it’s easier to get out of the ground and requires less capital. Rather than using the destructive processes of evaporation or mineral concentration, DLE selectively extracts lithium ions, either physically or chemically.
Eliminating the need for solar evaporation ponds, lime plants, and salt piles, direct lithium extraction from brine offers a more sustainable, faster process that results in higher quality lithium, in higher quantities.
IBAT’s proprietary DLE technology is the most sustainable, proven version of direct lithium extraction. Our facility is the first commercial facility to be built in North America and the world’s first modular facility. Our DLE technology gives producers speed to market, with lower CAPEX and OPEX.
Just as DLE is an improvement over traditional techniques, IBAT’s proprietary DLE technology via selective absorption using a proprietary crystal structure is a superior version of DLE technology.
Here’s how our technology has an edge over other DLE methods - ion exchange, solvent extraction, and membrane technology.
IBAT’s DLE Technology
vs Ion Exchange Methods
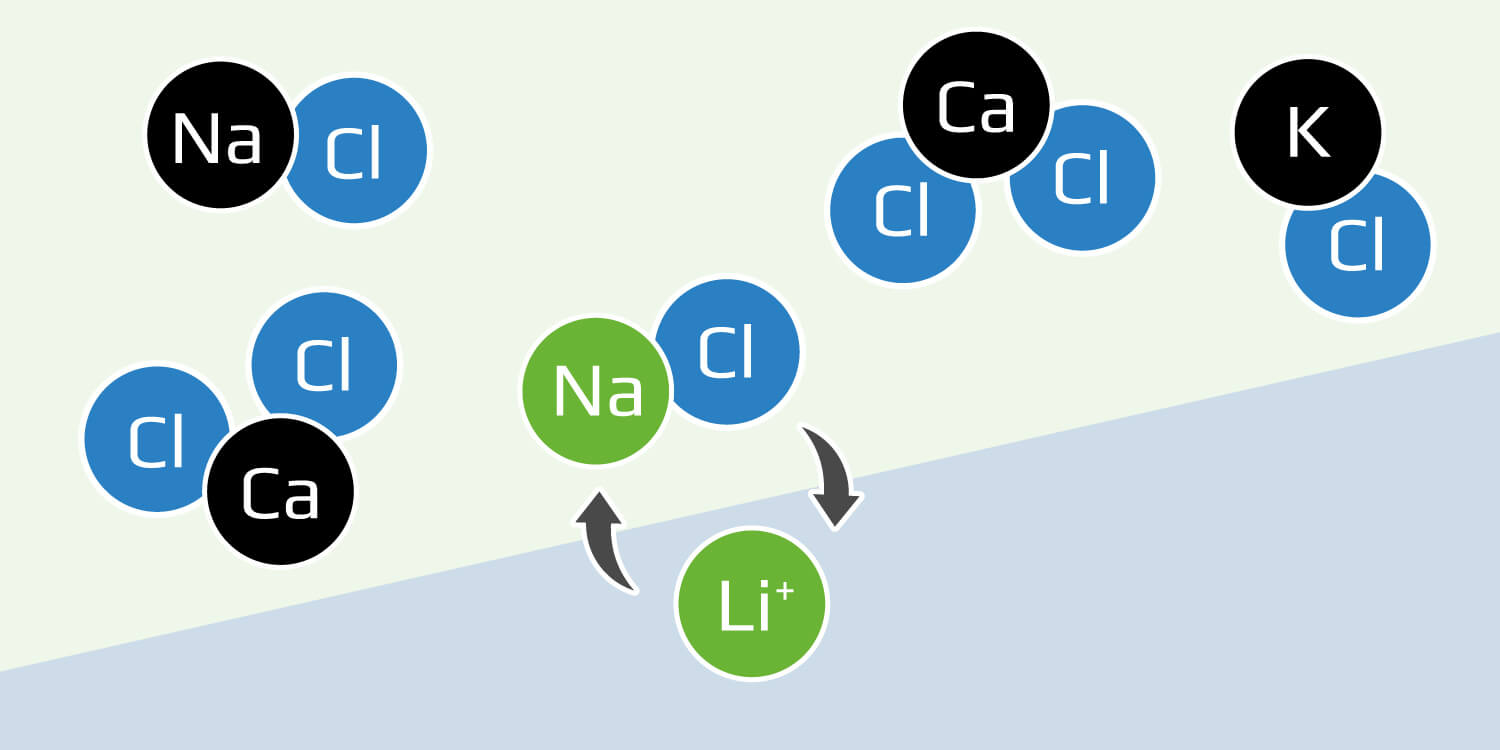
Ion exchange: A chemical reaction that uses ionic bonding, or the attraction between positively and negatively charged ions, to selectively pull lithium out of a solution and into tiny solids called resins.
Ion exchange, or IX, is already well-known to those in the mining industry primarily as a water purification technique. Beneficiation of copper, zinc, and iron ore, coal washing, and alumina refining all produce process water and wastewater that may be treated through IX. Many lithium producers are now turning to IX as a way to strip lithium out of brine.
In an ion exchange cycle, a brine solution is added to a bed of ion exchange resin made of a polymer and inorganic exchangers such as gel beads. The bed having been chemically treated to bind positively or negatively charged ions to it, when the solution is added, any ions in the brine that have a stronger affinity for the polymer than the ions already there will dislodge them and take their place in the bond. In this case, the lithium ions bond to the resin and the other elements are washed out.
To then remove the lithium from the system, the operator must treat it with acid. To regenerate the resin, the system must then be treated with base to return it to a neutral state. Not only does this process generate copious amounts of wastewater, little lithium is captured per cycle, because conventional IX resins have limited selectivity for lithium. So the cycle must be repeated multiple times, and each time generates more waste saltwater that must be dealt with.
How IBAT does it better:
- Our technology uses zero acids or bases, there’s no waste salt created.
- This makes our process both more economical and more environmentally friendly.
- With our process, the lithium extraction capacity doesn’t suffer due to the Gibbs-Donnan Effect–or unequal distribution of charged ions–when dealing with complex brines.
IBAT’s DLE Technology
vs Solvent Extraction Methods
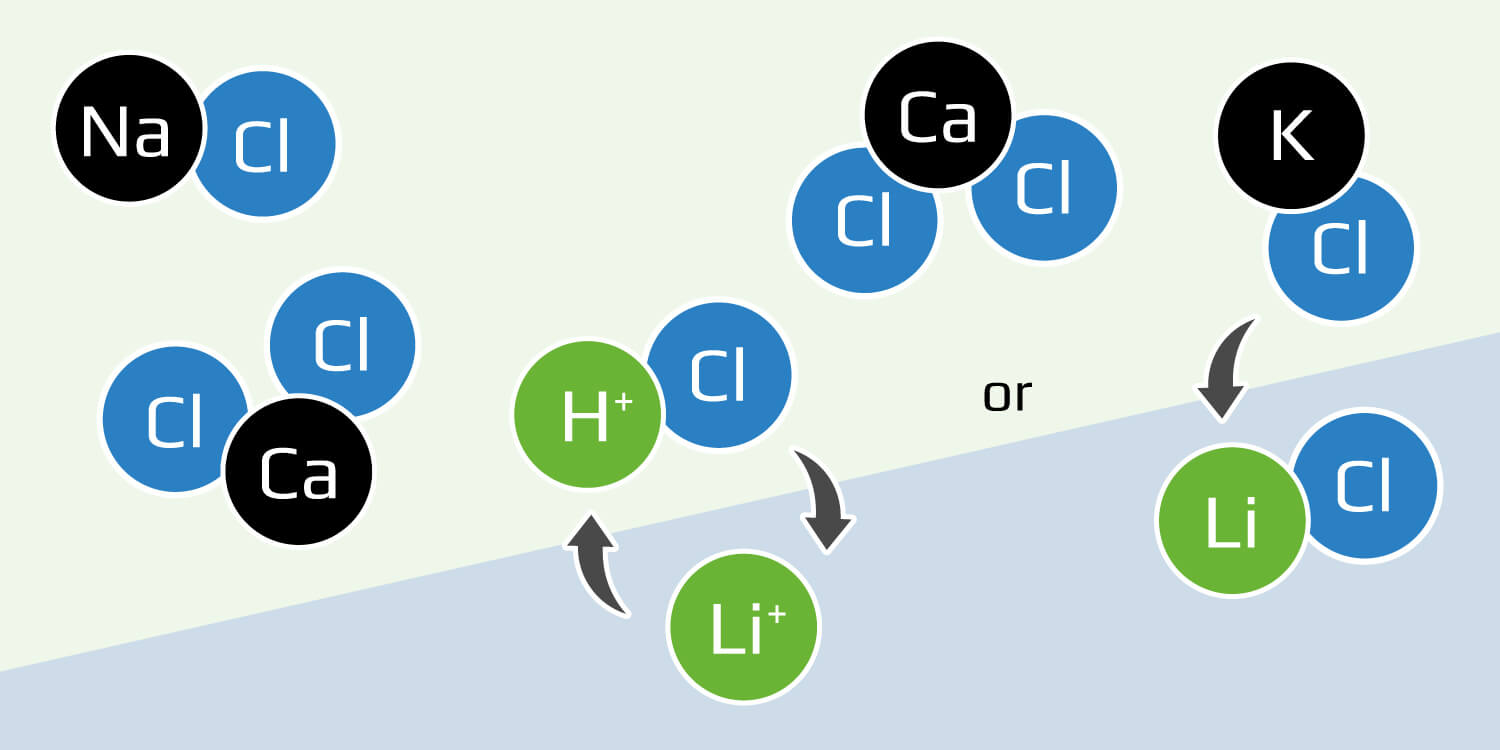
Solvent extraction: A separation method of placing the brine between two solvents that do not mix and shaking them together, causing the lithium to distribute into one layer and the magnesium and other impurities to transfer into the other layer.
Another common extraction technique is to utilize chelation or ion exchange between an organic solution with a solvent such as kerosene and an extractant, and an aqueous solution (water is the solvent). After the two immiscible liquids are combined, the lithium ions transfer to the organic phase, while most of the impurity metals remain in the aqueous phase. The organic phase must then be scrubbed, stripped with acid, then regenerated through acid-base neutralization.
Under lab conditions, solvent extraction produces excellent results on par with other DLE methods. However, when tested on solutions designed to mimic natural brine, the additional calcium, magnesium, zinc, and other elements can hinder the selectivity for lithium and render solvent extraction impractical unless the brine is pre-treated, an expensive step that also produces hazardous byproducts.
How IBAT does it better:
- Our process avoids the high volumes of harmful waste produced by solvent extraction’s scrubbing and stripping steps, as well as the corrosive solvents and process solutions required to make the process work.
- Our process is more economical, as it avoids the costs associated with expensive solvents, as well as their corrosive effect on costly equipment.
IBAT's DLE Technology
vs Membrane Technologies
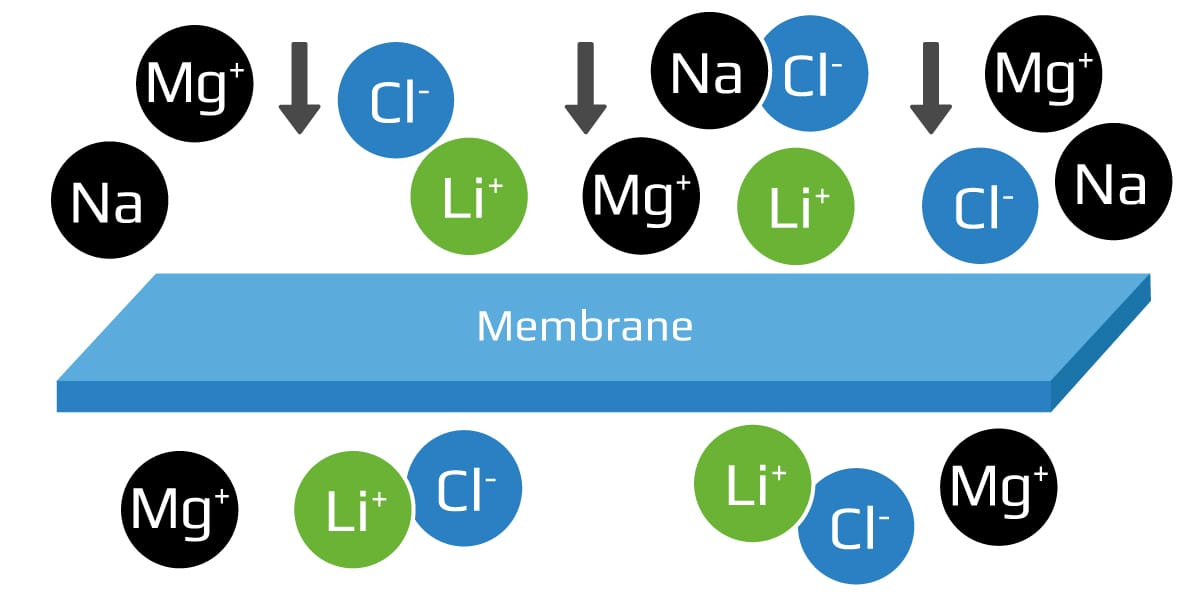
Membrane separation: Objective of the process is a purification method that uses a filter with tiny pores to remove as many impurities from the brine as possible before lithium extraction.
Semipermeable ceramic, polymer, or composite membranes are sometimes used as precursors to the lithium concentration stage to remove salt from chloride-rich brine. This may take the form of nanofiltration or reverse osmosis, in which pressure is used to push the brine across a filter to purify it, thereby reducing the operating costs and regeneration frequency of an IX system.
The shortcoming of membrane-based lithium extraction is that it’s only viable in salt concentrations that are much lower than those found in natural brines. Typically low lithium concentrations and high impurity concentrations are in the product stream. A natural brine will cause a membrane to quickly reach its treatment limit, at which point it ceases to remove impurities and can often rupture under the pressure. With nanofiltration systems, the brine must be pre-treated for the lithium to be separated effectively.
How IBAT does it better:
- IBAT’s DLE is able to handle the high levels of chloride, magnesium, and other elements present in natural brine without pretreatment and at scale.
With IBAT DLE,
the Environment is the Clear Winner
As we’ve seen, other extraction methods can wreak havoc on ecosystems. IBAT technology is the only lithium extraction process that could be called environmentally friendly. And with the demand for lithium only predicted to increase, now is the time to make it the industry standard. The planet can’t wait.
Work with IBAT
Call or email us today to learn how IBAT technology can be quickly deployed and brought online at your asset site.

